Rajkumar Savai is a professor at the Institute for Lung Health (ILH) of Justus Liebig University (JLU) in Giessen, Germany, where he leads the research group “Lung Microenvironmental Niche in Cancerogenesis.” He also heads the Oncology Basic Research Unit at Medical Clinic V of the University Hospital of Giessen and Marburg in Giessen. Moreover, he is jointly appointed as a group leader at the Department of Lung Development and Remodeling at the Max Planck Institute for Heart and Lung Research in Bad Nauheim, Germany.
Rajkumar Savai was born in Warangal, Telangana, India. After receiving his Masters (MSc) in Biochemistry from Kakatiya University, Warangal, India, he moved to Germany and finished his PhD and postdoctoral training at the Pneumology/Oncology Department of the JLU. In 2010, he moved to the Max Planck Institute for Heart and Lung Research in Bad Nauheim to run an independent junior research group. In 2020, he was appointed chair of Lung Microenvironmental Niche in Cancerogenesis professorship at the ILH, Giessen. He also serves as Scientific Coordinator at the German Center for Lung Research (DZL); Executive Committee Member of the Thoracic Oncology Assembly of the American Thoracic Society (ATS), Task Force Member of Lung Cancer–Translational Research for the national Network Genomic Medicine (nNGM) Lung Cancer, and Co-speaker of the LOEWE consortium ICANx (Cancer–Lung [Disease] Crosstalk: Tumor and Organ Microenvironment). Moreover, Dr. Savai is Principal Investigator of the excellence cluster at the Cardio-Pulmonary Institute (CPI) and is an associate member of the Frankfurt Cancer Institute (FCI). In 2018, he served as a Visiting Professor at the Earle A. Chiles Research Institute of Robert W. Franz Cancer Center at Providence Portland Medical Center in Portland, Oregon, USA. Dr. Savai has received several national and international awards for his scientific accomplishments. He has published 91 research articles (Date: 21th Sep 2021) in well-renowned scientific journals, including Nature Medicine, Science Translational Medicine, Science Advances, Nature Chemical Biology, Nature Communications, Circulation, Journal of Experimental Medicine, Journal of Clinical Investigation, Cancer Research, American Journal of Respiratory and Critical Care Medicine, and EMBO Molecular Medicine.
Dr. Savai´s lab aims to understand how the lung microenvironmental niche drives tumor initiation and progression and, most importantly, to identify specific targets that will interact with or inhibit the tumor niche’s maintenance and functional competence. The long-term goal of Dr. Savai´s lab is to translate tumor microenvironment-based therapeutic approaches into proof-of-concept clinical trials in lung cancer.
Lung cancer is the leading cause of cancer deaths worldwide. Although early-stage lung cancer can be treated with radical interventions or standard cytotoxic chemotherapy regimens, more than 70% of patients relapse and die, primarily due to metastatic progression. Moreover, most patients with stage IV cancer die within 18 months of diagnosis. Although recent clinical trials with immune checkpoint blockade therapy have shown an unprecedented durable response in patients with a variety of cancer, only a subset of patients achieves a stable state of cancer remission. Thus, further in-depth analysis of the molecular mechanisms underlying lung carcinogenesis is mandatory to identify novel molecular targets that may lead to the development of effective targeted therapies. Changes in the local lung microenvironment (niche) of transformed cells accompany malignant growth initiation. In response to cancer cell growth, the niche will evolve and modify its composition to create a complex network that supports the tumorigenic behavior and plasticity required for cancer cell growth, resistance to therapies, recurrence, and metastasis. Therefore, deeper knowledge of the niche establishment and evolution is needed to understand the complexity of cancer and to design better treatments.
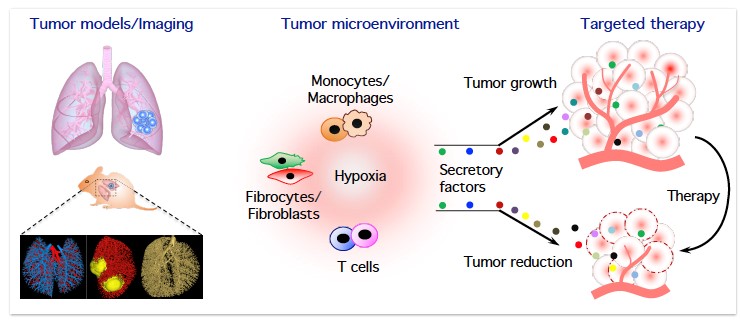
Figure 1: Overview of research topics addressed in Savai group
For the past several years, the Savai Lab is interested in several aspects of lung cancer, especially in the characterization of immune/inflammatory/mesenchymal cells in the tumor microenvironment and delineating the roles of monocytes/macrophages, fibrocytes/fibroblasts, and T cell subpopulations in lung cancer progression and metastasis. To investigate the above, the Savai Lab established several lung tumor mouse models (KRas, KRas/p53, and cRaf transgenic lung tumor models as well as primary and metastatic lung tumor models) and developed small animal lung tumor imaging techniques (micro-computed tomography and flat-panel volumetric computed tomography) to detect and monitor lung tumor growth and vascularization. Based on preliminary data of genome-wide profiling of human and mouse microenvironmental cells, the Professorship for Lung Microenvironmental Niche in Cancerogenesis focuses on the following major objectives:
Prof. Dr. Rajkumar Savai
Lung Microenvironmental Niche in Cancerogenesis
DZL Lung Cancer Disease Area Coordinator
Institute for Lung Health (ILH), Justus Liebig University Giessen
Aulweg 130
35392 Giessen
Max Planck Institute for Heart and Lung Research
Parkstr. 1
61231 Bad Nauheim
Tel: +49 (0) 641 99 46758
Tel: +49 (0) 6032 705 420
Fax: +49 (0) 6032 705 471
Email:
savai.rajkumar@innere.uni-giessen.de
rajkumar.savai@mpi-bn.mpg.de
Web:
https://www.mpi-hlr.de/68345/savai-lab
Monika Haselbauer
Assistentin
Tel: +49 (0) 6032 705 249
Fax: +49 (0) 6032 705 471
Email: Monika.Haselbauer@mpi-bn.mpg.de


Dr. Sascha Seidel, Sandra Medrano Garcia, Dr. Rajender Nandigama, Poonam Sarode, Dr. Kati Turkowski
Dr. med. Philipp Arndt, Joshua Ayoson, Dr. med. Michael Cekay, Öznur Cetin, Annika Karger, Dr. vet med. Siavash Mansouri, Spyridoula Barmpoutsi, Joshua Olapoju, Selma Dizdarevic, José Alonso
Jeanette Knepper, Yanina Knepper
Alina Asafova, David Brunn, Katharina Ebel, Nefertiti El-Nikhely, Tamara Haselbauer, Marianne Hoeck, Stefanie Pfrogner, Ylia Salazar, Anja Schmall, Samantha Storn, Prema Subarayal, Aleksandra Tretyn, Natasha Vipotnik, Xiang Zheng